PCR Testing in Minutes with Digital Microfluidics
Posted 2/9/2021 (Updated 4/5/2021)
According to the CDC, the “gold standard” for SARS-CoV-2 testing is reverse transcription polymerase chain reaction (or RT-PCR). The PCR method makes many copies of a small strand of DNA by thermal cycling: DNA double strands are denatured at a high temperature (typically 95ºC) followed by binding of primers to the target DNA of interest and extension of the DNA strands at a lower temperature (typically 60-65ºC). This process is repeated to exponentially make millions of copies of DNA enabling optical detection via fluorescence.
How Digital Microfluidics Enables Ultra-rapid PCR Results
Digital microfluidics (DMF) technology miniaturizes RT-PCR protocols using standard laboratory chemistries – but fully automated. Low reaction volumes enable rapid thermal cycling due to low thermal mass which helps speed up PCR. Our DMF platform has demonstrated PCR within 5 minutes through optimization of the following factors:
Low Droplet Volume
Digital microfluidics programmably manipulates discrete microliter-sized droplets of reagents and samples enabling ultra-rapid heating and cooling, which is essential for PCR assays. In DMF, PCR is performed by shuttling tiny droplets of reaction mixtures between two heat zones that are maintained at ~95ºC and ~60ºC as required by the PCR assays.
This is essentially mimicking the original PCR method as invented by Dr. Mullis, where the reaction mixture was transported between two heat baths. Microliter-sized droplets have low thermal mass and can be heated and cooled with no lag time; this can be performed at a very rapid rate if the heaters and sensors are in immediate contact with the droplets.
Built-in Heaters and Sensors on Cartridge
Digital microfluidic protocols for PCR have the heaters and sensors embedded within the disposable cartridge abutting the droplets to further allow rapid thermal cycling.
Generally with other PCR platforms, heating and cooling are performed conductively with a heat block or convectively adding additional thermal mass which slows down the assay considerably in comparison to DMF. Further in DMF cartridges, heating zones are localized just to the microliter droplet further reducing the thermal mass.
Easy Sample Preparation and Microliter-sized Reagents
In conjunction with ultra-rapid PCR, sample prep needs to be fast, too. Typically, respiratory and other physiological specimens require sample preparation to lyse the pathogens to release the RNA. This is further captured onto a solid phase such as magnetic beads.
Digital microfluidics allows for automatic and rapid sample preparation methods and enables concentration of all the RNA – captured on the magnetic beads, into a single droplet – which can then be eluted off into another droplet to perform RT-PCR.
DMF requires only miniscule amounts of reagents and, therefore, supply scarcities would only have a muted impact on the number of tests that could be performed. This is particularly beneficial during the current COVID-19 pandemic as reagent supplies are unpredictable.
Feasibility of PCR Results in 5 Minutes using DMF
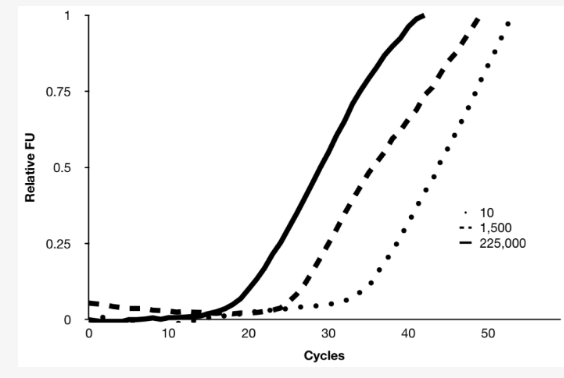
Baebies scientists have published several peer-reviewed publications on PCR using DMF technology. In this 2020 peer-reviewed publication in Diagnostics, PCR for congenital cytomegalovirus (cCMV) infection was performed on a DMF platform and cartridge within 5 minutes.
Table: CMV PCR assay using a prototype DMF cartridge. Successful amplification of CMV (10, 1,500, and 225,000 copies/µL) is achieved within 5 minutes. FU: fluorescence units.
Related Links:
- Baebies Receives FDA Breakthrough Device Designation for First Point-of-Care Heparin Monitoring Test
- Learn more about Digital Microfluidic Technology
- Baebies Expands Its Vision: Any Test, Anywhere, Everyone™
- Feasibility of PT, aPTT, and Fibrinogen on a Multifunctional Digital Microfluidic Cartridge
- Novel Fluorometric Assay for Pediatric Anti-Factor Xa Testing: Minimizing Bilirubin and Hemolysis Interference in Whole Blood